Additional information
| Qty | 100 μg, 500 μg, 1 mg, 5 mg, 10 mg |
|---|---|
| Shipping in Dry Ice | yes |
170,00€ – 4.400,00€
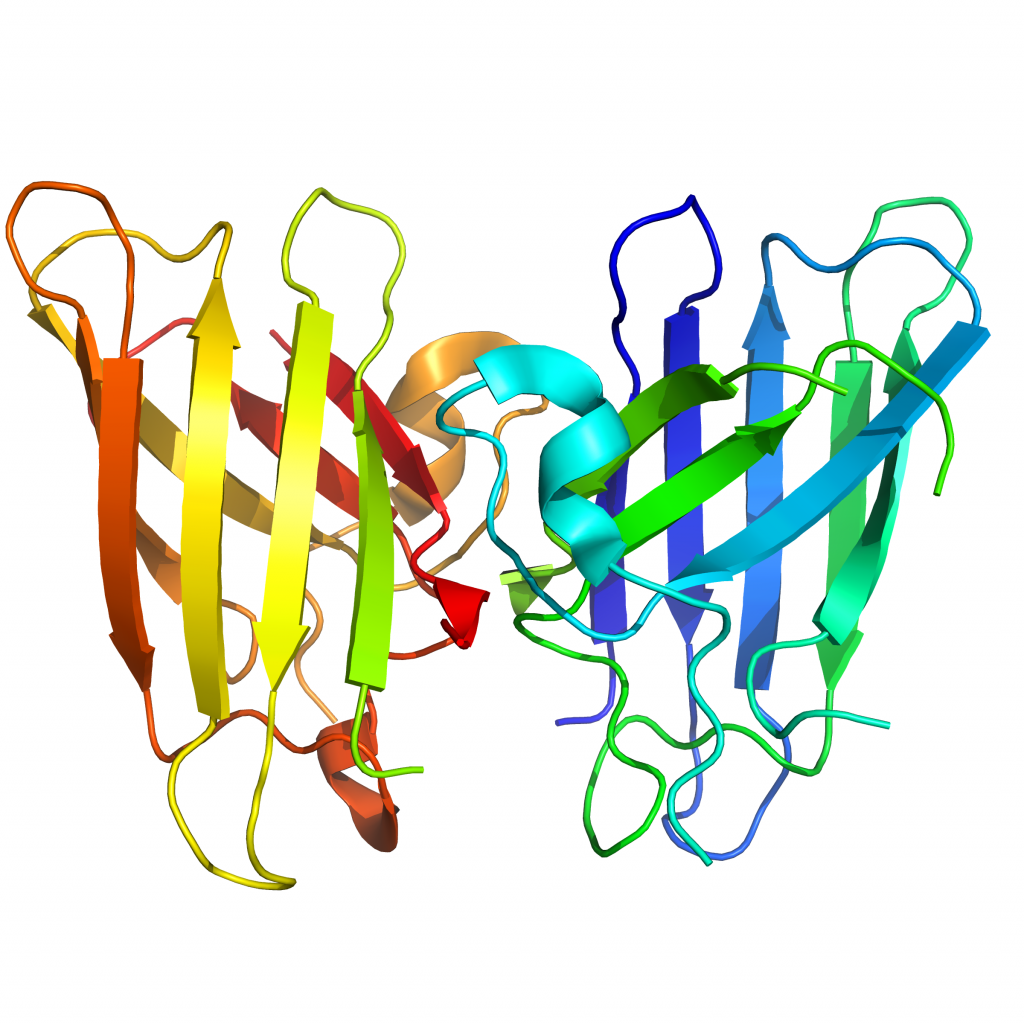
Human, recombinant
Full length, UniProtKB accession P00441
MW = 32600 Da (dimer)
EC # 1.15.1.1.
CAT # G06SD001
| Catalog n. | Qty | Price |
|---|---|---|
| 170,00€ | ||
| 520,00€ | ||
| 840,00€ | ||
| 2.600,00€ | ||
| 4.400,00€ | ||
| VAT not included | ||
For any special request or bulk quantities Click Here
| Qty | 100 μg, 500 μg, 1 mg, 5 mg, 10 mg |
|---|---|
| Shipping in Dry Ice | yes |
Sequence
10 20 30 40 50
GSFT-MATKAVCVLK GDGPVQGIIN FEQKESNGPV KVWGSIKGLT EGLHGFHVHE
60 70 80 90 100
FGDNTAGCTS AGPHFNPLSR KHGGPKDEER HVGDLGNVTA DKDGVADVSI
110 120 130 140 150
EDSVISLSGD HCIIGRTLVV HEKADDLGKG GNEESTKTGN AGSRLACGVI
GIAQ
Available mutant
*Amyotrophic lateral sclerosis (ALS) related.
By continuing to use the site, you agree to the use of cookies. more information
The cookie settings on this website are set to "allow cookies" to give you the best browsing experience possible. If you continue to use this website without changing your cookie settings or you click "Accept" below then you are consenting to this.